Study design and population
We conducted a cross-sectional, observational study in the EpiChron Cohort (Prados-Torres et al., 2018). This cohort links demographic and clinical anonymized information based on real-world data from the electronic health records (EHRs) and clinical-administrative databases of public health system users of the Spanish region of Aragón (1.3 M inhabitants). We included in this study all individuals aged 40 years and older on January 1, 2015 (n= 673,059), and we selected those patients with a diagnosis of COPD and/or HF in their primary and/or hospital care EHRs. We stratified the study population by sex and as having COPD (n= 28,608), HF (n= 13,414), or COPD+HF (n= 3,952). The Clinical Research Ethics Committee of Aragón (CEICA) approved the research protocol of this study (PI16/0136) and waived the requirement to obtain informed consent from patients since the information used was anonymized.
Study variables
For each patient, we studied their sex, age, and all the chronic conditions registered in their primary and/or hospital care EHRs in the index date. Diseases were coded according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM), or the International Classification of Primary Care (ICPC), and then grouped into 260 mutually exclusive Expanded Diagnostic Clusters (EDCs) through the Johns Hopkins ACG System (version 11.0, The Johns Hopkins University, Baltimore, MD, US). We included in the study the list of 114 EDCs defined as chronic in the study by Salisbury et al. (Salisbury et al., 2011), which has been validated in previous studies on multimorbidity (Poblador-Plou et al., 2014; Prados-Torres et al., 2012). Comorbidities were included in the analysis as binary variables (i.e., absence/presence).
Characteristics of the study population
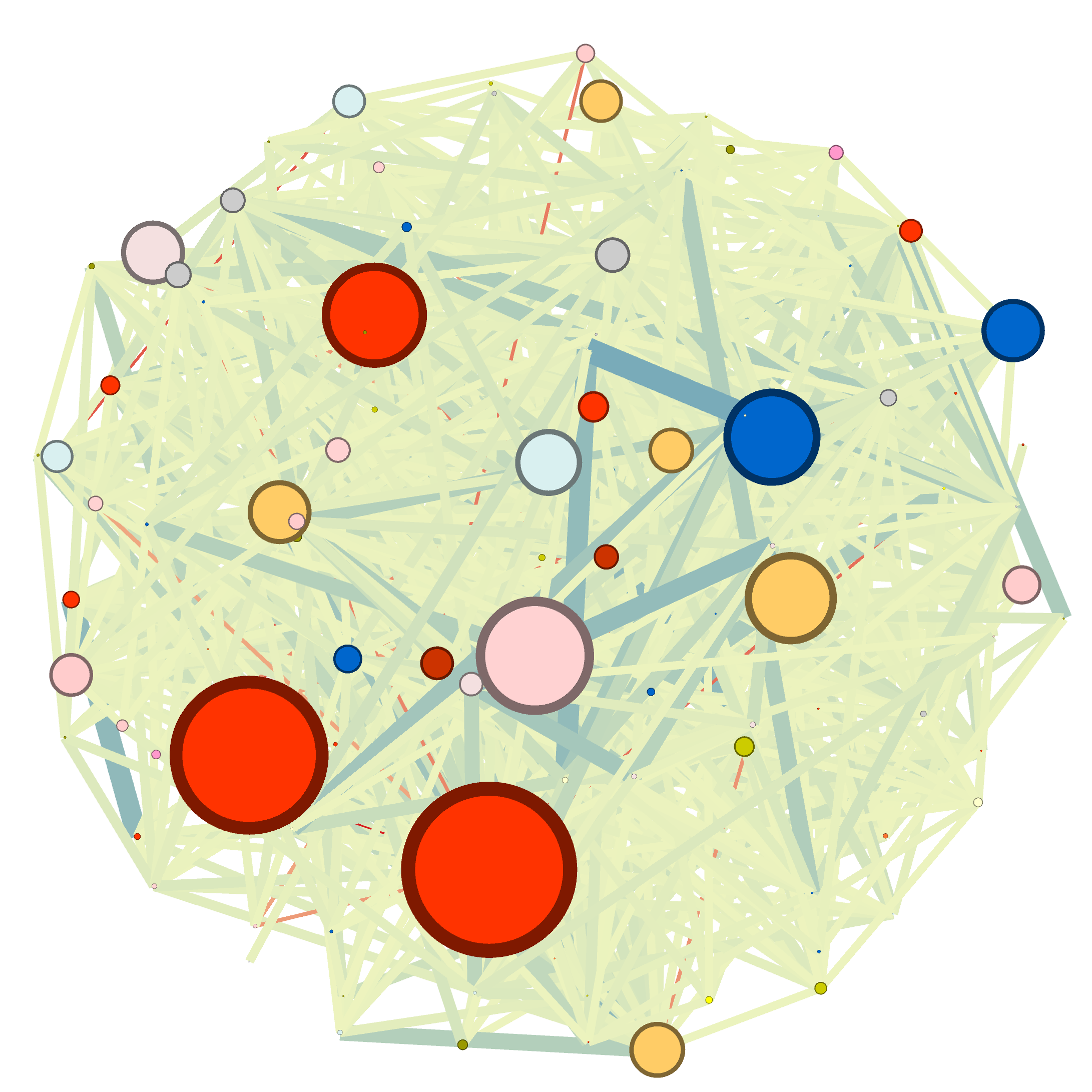
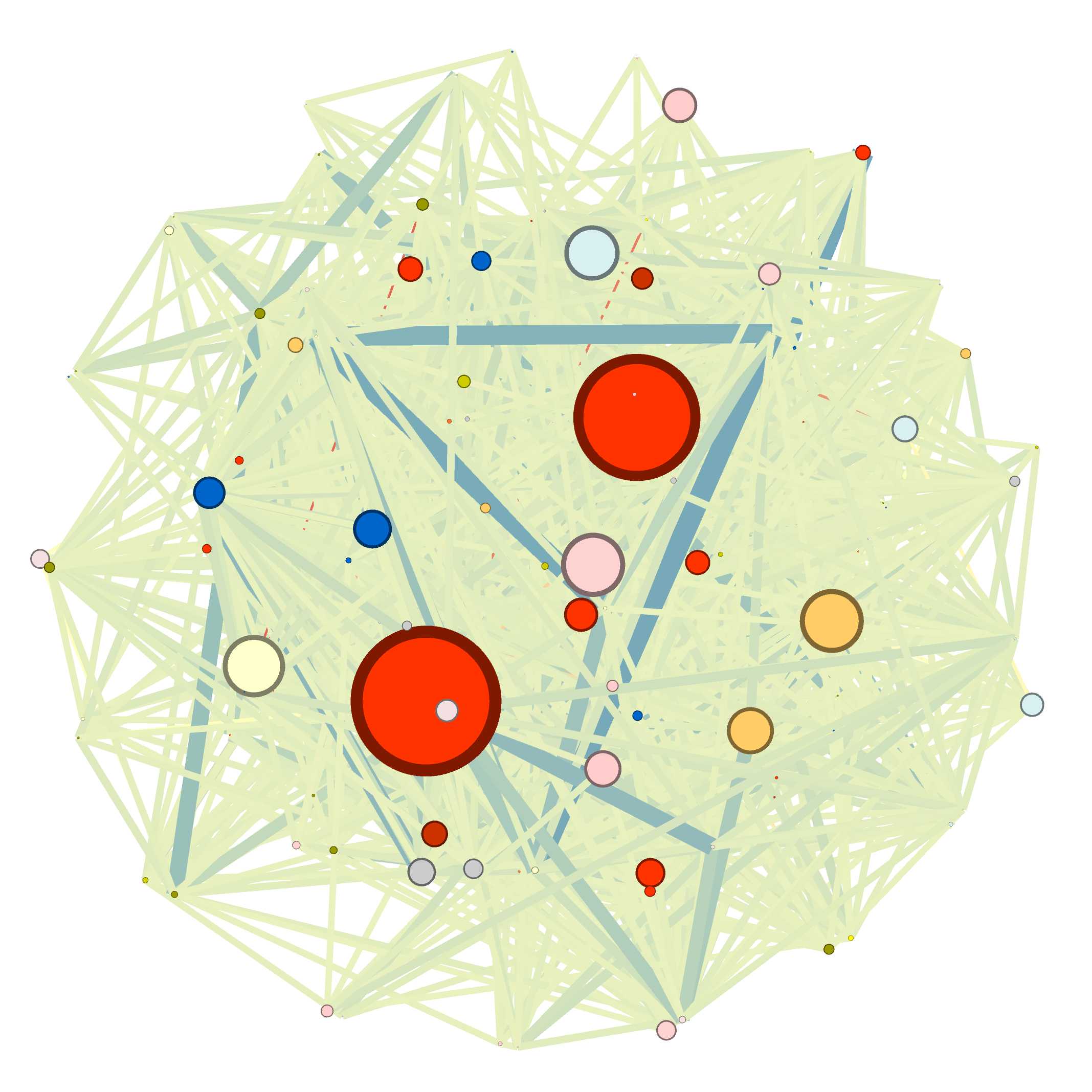
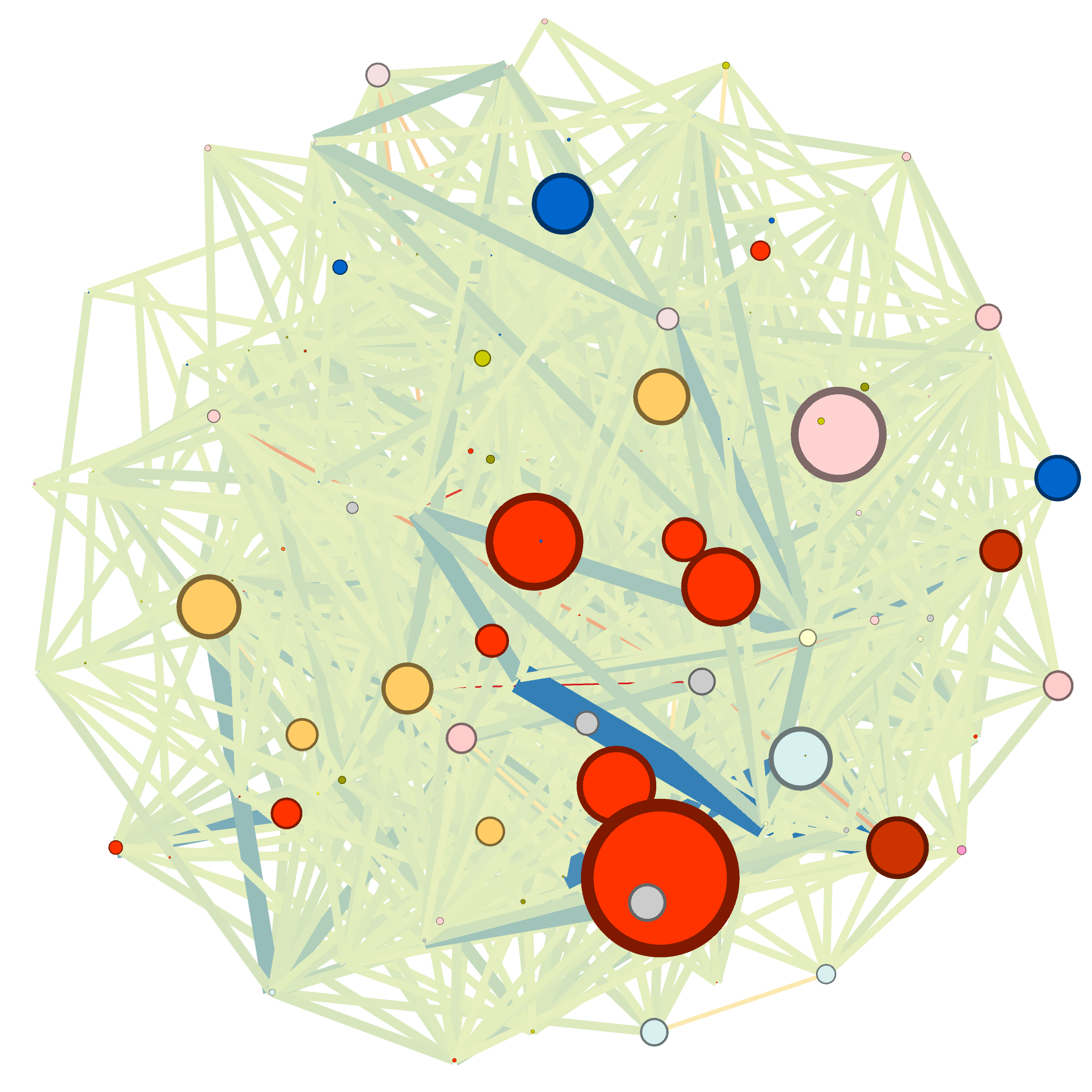
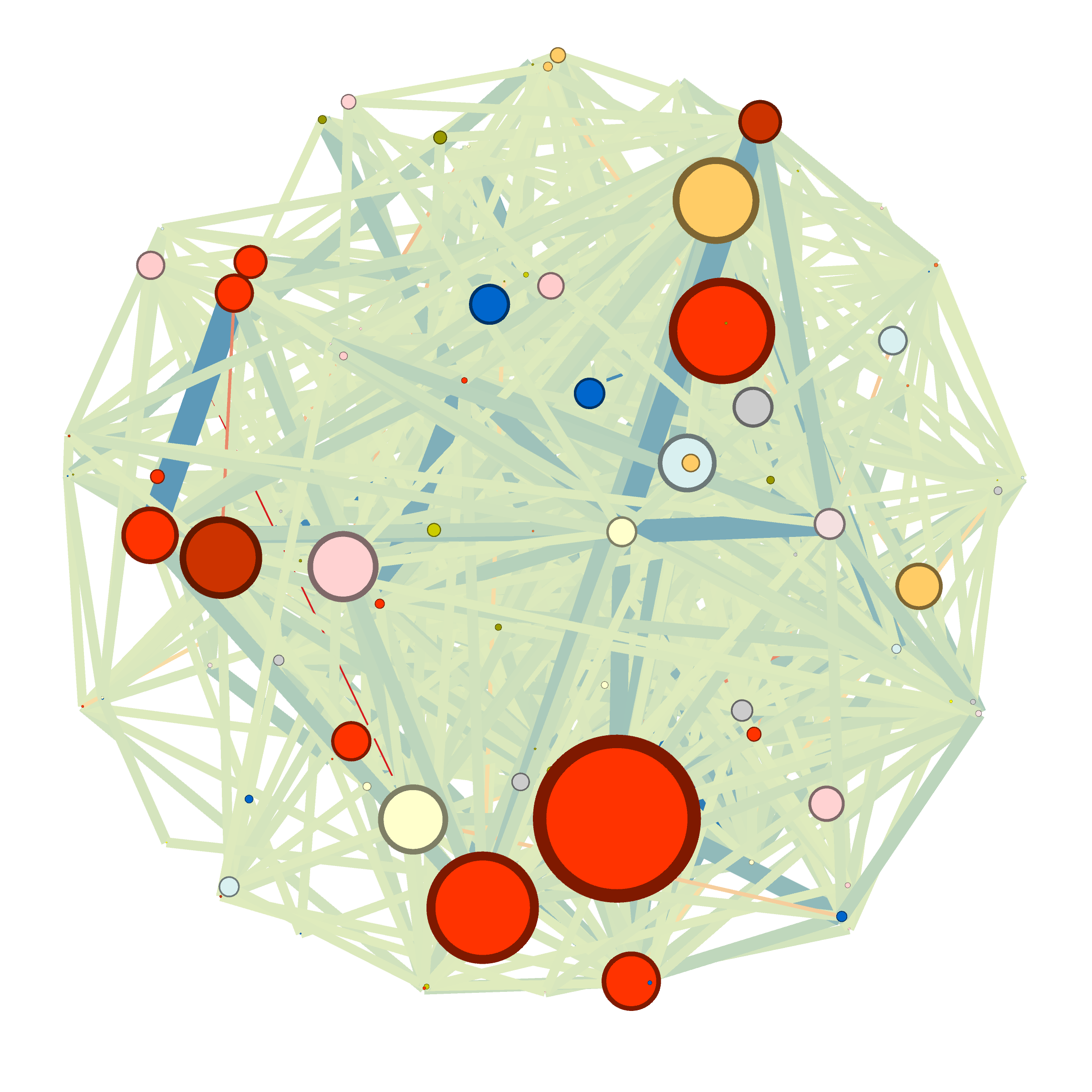
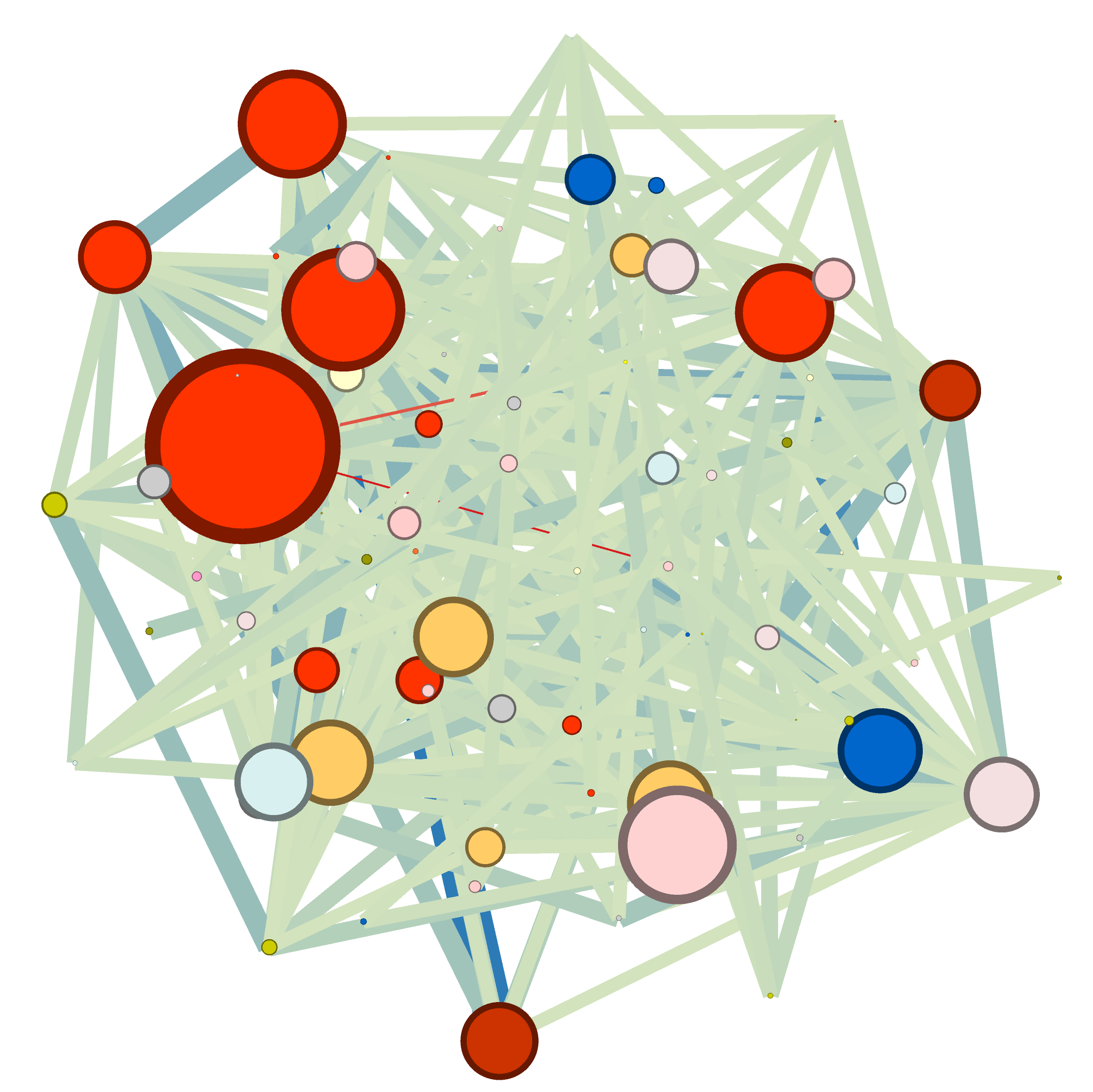
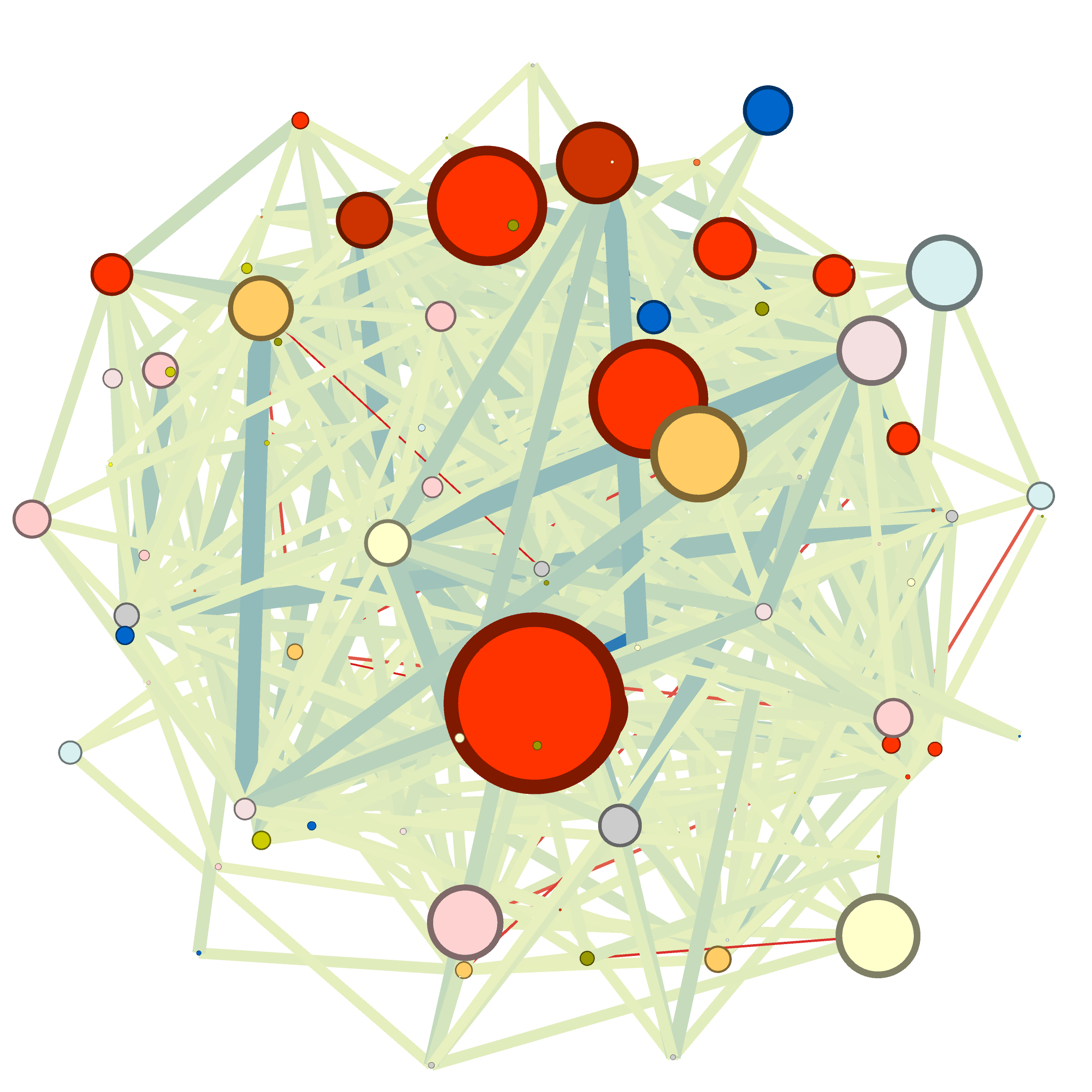
In the population 40 years of age and older of the EpiChron Cohort, the prevalence of COPD, HF and COPD+HF was 4.25%, 1.99%, and 0.59%, respectively. The demographic characteristics with each index condition (COPD, HF, or COPD+HF) are shown above. Women were almost twice as frequent than men in the population with HF (64% vs. 32% and 37% in the COPD and COPD+HF groups, respectively). The HF group was on average 10 years older than the COPD group (80.3 ± 11.0 vs. 70.7 ± 12.1 years) and presented a higher mean number of comorbidities (8.1 vs. 5.4 conditions). Hypertension, dyslipidemia, arthritis, and diabetes mellitus appeared within the most frequent chronic conditions in all groups.